Chemistry in the Kitchen: Baking Soda vs. Baking Powder (and a recipe for pancakes too!)
/Do you or your kids ever wonder what makes your pancakes fluffy? Or cupcakes rise? In today’s after-school program, we’re going to do a couple of science experiments to see what baking soda and baking powder do. Then we’re going to make pancakes! Your kids can easily recreate the experiment and our recipe in your kitchen at home! These days, this is about as close as I come to using my degree in Chemical Engineering :)
Banana bread, chocolate cake, and your birthday party cupcakes come out of the oven way higher than when they started. Why? If you’re not using yeast, there’s a good bet the recipe included baking soda and/or baking powder, two very common leavening agents.
BAKING SODA
Baking soda is just sodium bicarbonate, which is a base. In the presence of moisture, it reacts with acid to produce carbon dioxide, which are the little bubbles you see forming in your batter. The little bubbles trap air inside your batter, causing the batter to become “fluffier”. The bubbles are trapped in there forever! Many times, you will see baking soda used in recipes with buttermilk, which is a very common acid. Have you ever been told not to add the dry ingredients to the liquid ingredients until the very end of a recipe? That’s because as soon as the dry and wet ingredients are combined, the chemical reaction between the base (baking soda) and acid (buttermilk) will begin! If you don’t hurry up and start cooking the food, all of that carbon dioxide that you produce will fizzle out. You want to start cooking while all of those little bubbles are trapped inside your batter!
BAKING POWDER
Baking powder is made with sodium bicarbonate PLUS cream of tarter and cornstarch. Why those 2 additional ingredients? The cream of tarter is a dry acid. The cornstarch keeps any moisture at bay - since moisture is what starts the chemical reaction. So baking powder doesn’t require an acid to work, since it’s all-in-one. It will start to work on it’s own when it comes in contact with moisture. Most baking powder is “double-acting” so it will create carbon dioxide (those little bubbles) immediately after coming into contact with moisture, and again during heating. This longer-lasting reaction usually creates fluffier baked goods than baking soda alone.
So why wouldn’t you just use baking powder? Baking soda has some great side-effects. It will help give your food a nice brownish color. Also, when combined with an acid, it will create a more effective initial reaction than with baking powder. Just don’t use it without an acid, or your food might taste a bit like metal!
EXPERIMENT: Are you ready to see how it works?
Equipment & ingredients:
Measuring cup
Teaspoon
Clear Cups or test tubes if you want to get all science-y!
Water
Vinegar
100% juice
Stopwatch or timer
Directions:
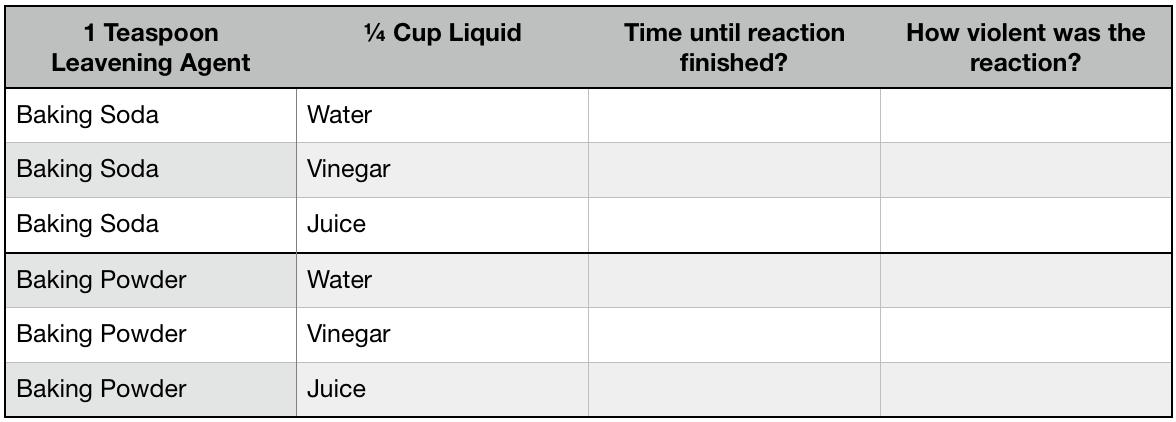
You will test baking soda in each of the 3 liquids: water, vinegar, and juice. Then you will test baking powder in each of the 3 liquids: water, vinegar, and juice.
Fill a clear cup with ¼ cup of water. Add 1 teaspoon baking soda and start the timer. Finish timing when all the little bubbles stop rising to the surface. Record your answer.
Repeat with remaining 5 iterations: vinegar/baking soda, juice/baking soda, water/baking powder, vinegar/baking powder, juice/baking powder.
Questions:
What reacted the most violently?
Which reactions lasted the longest?
A picture of our experiment. Can you guess which set is with baking soda and which set is with baking powder?
RECIPE: Now, go make some pancakes! Here’s an easy recipe that I’ll be using with my class today:
Ingredients
2 cups flour
2 eggs
1 cup milk
1 cup buttermilk
½ teaspoon baking soda
2 teaspoons baking powder
1 teaspoon salt
1 tablespoon sugar
Directions
Heat a griddle to medium-high heat.
Crack eggs into a small bowl and remove any shells. Mix with a fork.
Measure dry ingredients (flour, baking soda, baking powder, salt, sugar) into a bowl and whisk to combine.
Measure wet ingredients (egg, milk, buttermilk) into a bowl and stir to combine.
Combine wet and dry ingredients and stir until just mixed.
Measure out ¼ cup per pancake on griddle. Flip when the edges move away from the pan and bubbles form on top.
Cook for 1 more minute.
EAT!